Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 H monitoring in radioactive gaseous waste
H monitoring in radioactive gaseous wasteFurutani, Misa; Kometani, Tatsunari; Nakagawa, Masahiro; Ueno, Yumi; Sato, Junya; Iwai, Yasunori*
Hoken Butsuri (Internet), 55(2), p.97 - 101, 2020/06
Herein, an oxidation catalyst was introduced after heating it to 600 C to oxidize tritium gas (HT) existing in exhaust into tritiated water vapor (HTO). This study aims to establish a safer
C to oxidize tritium gas (HT) existing in exhaust into tritiated water vapor (HTO). This study aims to establish a safer  H monitoring system by lowering the heating temperature required for the catalyst. In these experiments, which were conducted in the Nuclear Science Research Institute, Japan Atomic Energy Agency, cupric oxide, hydrophobic palladium/silicon dioxide (Pd/SiO
H monitoring system by lowering the heating temperature required for the catalyst. In these experiments, which were conducted in the Nuclear Science Research Institute, Japan Atomic Energy Agency, cupric oxide, hydrophobic palladium/silicon dioxide (Pd/SiO ), and platinum/aluminum oxide (Pt/Al
), and platinum/aluminum oxide (Pt/Al O
O ) catalysts were ventilated using standard hydrogen gas. After comparing the oxidation efficiency of each catalyst at different temperatures, we found that the hydrophobic Pd/SiO
) catalysts were ventilated using standard hydrogen gas. After comparing the oxidation efficiency of each catalyst at different temperatures, we found that the hydrophobic Pd/SiO and Pt/Al
and Pt/Al O
O catalysts could oxidize HT into HTO at 25
catalysts could oxidize HT into HTO at 25 C.
C.
Irisawa, Keita; Kudo, Isamu*; Taniguchi, Takumi; Namiki, Masahiro*; Osugi, Takeshi; Nakazawa, Osamu
QST-M-16; QST Takasaki Annual Report 2017, P. 63, 2019/03
no abstracts in English
Sato, Junya; Suzuki, Shinji*; Kato, Jun; Sakakibara, Tetsuro; Meguro, Yoshihiro; Nakazawa, Osamu
QST-M-2; QST Takasaki Annual Report 2015, P. 87, 2017/03
no abstracts in English
Nagaishi, Ryuji; Motooka, Takafumi; Yamagishi, Isao
Proceedings of 2016 EFCOG Nuclear & Facility Safety Workshop (Internet), 6 Pages, 2016/09
Overflow of water from waste storage tanks of High Integrity Containers (HIC) in the multi-nuclide removal equipment (ALPS) was discovered at Fukushima Daiichi NPS in April of last year. The mechanism of overflow was not understood very much at that time. To elucidate that for chemical safety in the waste storage, irradiation experiments of simulated carbonate slurry by Co-60  -rays have been conducted in CLADS, JAEA in cooperation with TEPCO, TOSHIBA and KURITA. Hydrogen molecule was the main radiolytic gas product in the slurry, and its amount was enhanced by dissolved species of not only halide ions as seawater components but also carbonate ion as an additive for co-precipitation at a basic condition. The bubbles of molecules were further formed and almost held in the slurry without stirred. These sequentially led to the expansion of slurry, and then to its separation into the shrunk one and supernatant water, which was little accumulated without irradiated.
-rays have been conducted in CLADS, JAEA in cooperation with TEPCO, TOSHIBA and KURITA. Hydrogen molecule was the main radiolytic gas product in the slurry, and its amount was enhanced by dissolved species of not only halide ions as seawater components but also carbonate ion as an additive for co-precipitation at a basic condition. The bubbles of molecules were further formed and almost held in the slurry without stirred. These sequentially led to the expansion of slurry, and then to its separation into the shrunk one and supernatant water, which was little accumulated without irradiated.
Taniguchi, Naoki; Kawasaki, Manabu*; Fujiwara, Kazuo*
JNC TN8400 2001-011, 62 Pages, 2001/03
The corrosion of metallic materials used in natural environment are sometimes affected by microbial action. It is apprehended that microorganism living in deep underground or brought from ground surface during excavation makes an impact on overpack material for geological disposal of high-level radioactive waste. Sulfate reducing bacteria (SRB) is known to be one of the most representative microorganism which affects the corrosion of metals. In this study, the behavior of growth of SRB was investigated at first under the presence of bentonite as a main component of buffer material which encloses the overpack. The results of the tests showed that the population of SRB after the culture in synthetic sea water mixed with bentonite decreased with increasing the ratio of bentonite/solution. SRB was hardly grown in medium whose bentonite/solution ratio exceeded 1000g/l. As a conservative case, the effects of sulfide on the corrosion of overpack materials were also studied assuming high activity of SRB. Carbon steel, copper and titanium specimens were immersed in synthetic sea water purging 0.1MPa H S gas and the corrosion behavior was compared with the results in N
S gas and the corrosion behavior was compared with the results in N gas purging environment. Obvious effect of sulfide on the corrosion of carbon steel was not observed, but the corrosion rates of copper specimens were accelerated several hundred times by purging H
gas purging environment. Obvious effect of sulfide on the corrosion of carbon steel was not observed, but the corrosion rates of copper specimens were accelerated several hundred times by purging H S gas. The absorption of hydrogen into titanium specimens was not affected by purging H
S gas. The absorption of hydrogen into titanium specimens was not affected by purging H S gas, but the difference of hydrogen absorption between pure titanium and titanium alloy containing 0.06%-Pd was observed.
S gas, but the difference of hydrogen absorption between pure titanium and titanium alloy containing 0.06%-Pd was observed.
Tanai, Kenji; Sato, Haruo; *; *
JNC TN8400 99-045, 108 Pages, 1999/11
In the anaerobic environment in the deep underground water, carbon-steel overpack corrodes and generates molecular hydrogen. It is conceivable that this hydrogen either dissolves into the porewater of the buffer and migrates through the buffer. If the rate of aqueous diffusion of hydlogen is too low compared to the rate of hydrogen generation, the concentration of hydrogen at the overpack surface will increase until a solubility limit is attained and a free hydrogen gas phase forms. It is possible that the pressure in this accumulating gas phase will increase, affecting the stability of the buffer or the surrounding rock mass. There is also a concern of possible effects on nuclide migration, as it is also conceivable that the flow of gas could push out radionuclide-bearing porewater in the buffer when it floes through the buffer. As such, experimental and analytical study must be carried out on such phenomenon to evaluate such potential phenomena. (1)Diffusion experiment of dissolved hydrogen. According to the test result concerning the effective diffusion coefficient of the dissolved hydrogen in buffer material, the effective diffusion coefficient of reference buffer material (70wt% bentonite + 30wt% sand mixture, dry density 1.6Mg m ) ranges from 10
) ranges from 10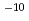 m
m s
s to 10
to 10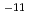 m
m s
s . The value of the effective diffusion coefficient measured for a dry density of 1.8 Mg m
. The value of the effective diffusion coefficient measured for a dry density of 1.8 Mg m is slightly smaller than the value in that for a dry density 1.6 Mg m
is slightly smaller than the value in that for a dry density 1.6 Mg m . And the effective diffusion coefficient at 60
. And the effective diffusion coefficient at 60 C tends to have slightly larger value than that at 25
C tends to have slightly larger value than that at 25 C. Test results from the foreign countries show the diffusion coefficient in the range between 10
C. Test results from the foreign countries show the diffusion coefficient in the range between 10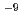 m
m s
s to 10
to 10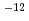 m
m s
s . Basically, these test results reported here are in the same range as these other results. (2)Gas permeability. Studies of the gas permeabinty of buffer material have been carried out by Pusch et al., Volckaert ...
. Basically, these test results reported here are in the same range as these other results. (2)Gas permeability. Studies of the gas permeabinty of buffer material have been carried out by Pusch et al., Volckaert ...
Hashizume, Shuji; Matsumoto, Junko; Bamba, Tsunetaka
Zairyo To Kankyo, 47(10), p.638 - 644, 1998/00
no abstracts in English
Wada, Ryutaro*; Nishimura, Tsutomu*; Fujiwara, Kazuo*; *; *
PNC TJ1058 97-003, 33 Pages, 1997/03
None
Tokunaga, Okihiro; Aoki, Yasushi; Sato, Shoichi; *; *; *; *
Applications of Isotopes and Radiation in Conservation of the Environment, p.105 - 114, 1992/00
no abstracts in English
; *; *
Japanese Journal of Applied Physics, 19(9), p.1703 - 1710, 1980/00
Times Cited Count:10 Percentile:48.16(Physics, Applied)no abstracts in English
; Nekoya, Shinichi; Emori, Kouichi; Ogawa, Masuro; Ouchi, Mitsuo; Okamoto, Yoshizo; Sanokawa, Konomo; *; *; *; et al.
Nihon Genshiryoku Gakkai-Shi, 22(3), p.181 - 188, 1980/00
Times Cited Count:1 Percentile:22.79(Nuclear Science & Technology)no abstracts in English
Hishida, Makoto; Takizuka, Takakazu; Ogawa, Masuro; Nekoya, Shinichi; Emori, Kouichi; Ouchi, Mitsuo; Sanokawa, Konomo
Nihon Genshiryoku Gakkai-Shi, 22(5), p.326 - 334, 1980/00
Times Cited Count:2 Percentile:33.85(Nuclear Science & Technology)no abstracts in English
; Ando, Yoshio*
Yosetsu Gakkai-Shi, 32(2), p.91 - 100, 1963/00
no abstracts in English
Sato, Junya; Suzuki, Shinji; Nakagawa, Akinori; Kato, Jun; Sakakibara, Tetsuro; Meguro, Yoshihiro; Nakazawa, Osamu
no journal, ,
no abstracts in English
Nagaishi, Ryuji; Motooka, Takafumi; Yamagishi, Isao; Inoue, Masao; Matsumura, Taichi
no journal, ,
no abstracts in English
Arai, Yoichi; Hinai, Hiroshi; Koma, Yoshikazu; Ikeda, Akira*; Obata, Masamichi*; Shibata, Atsuhiro; Nomura, Kazunori
no journal, ,
no abstracts in English
Yoshida, Ryoichiro; Yamane, Yuichi; Abe, Hitoshi
no journal, ,
At criticality accident on solution fuel, it is known that hydrogen gas is generated with nuclear fission. Because of the temporary decrease effect on power through density decrease and the possibility which causes secondary accident such as explosion, it is important on safety evaluation at criticality accident that to estimate relating the amount of hydrogen gas and the number of nuclear fissions. The time history data of concentration of hydrogen gas in vent gas when the transition critically used uranyl nitrate solution, which was obtained from Transient Experiment Critical Facility (TRACY), could not be explained by simple diffusion, because of peak about 500 seconds later from reactivity addition and after that long tail. Then, calculating the transition behavior of hydrogen gas in reactor core tank by combining multiple models such as Taylor dispersion, and evaluating concentration of hydrogen gas result reproducing the measured value well. The amount of hydrogen gas also evaluated 4 mol per 10 fissions.
fissions.
 gas production by radiolysis of carbonate slurry generated from Multi-Radionuclide Removal System
gas production by radiolysis of carbonate slurry generated from Multi-Radionuclide Removal SystemArai, Yoichi; Hinai, Hiroshi; Koma, Yoshikazu; Shibata, Atsuhiro; Nomura, Kazunori
no journal, ,
Multi-Radionuclide Removal System (MRRS) has been utilized for decontaminate of the radioactive contaminated water in Fukushima Daiichi Nuclear Power Station (1F). The secondary wastes including iron hydroxide, carbonate and used adsorbents are packed in the High Integrity Container (HIC). It was reported that the radioactive liquid was overflowed in several HICs. This phenomenon was only observed in HICs containing the carbonate slurry waste. It was inferred that volume increase of wastes due to gas generation caused by the water radiolysis. However, the gas generation behavior was not well known. Therefore, it is important to investigate gas generation behavior of radioactive carbonate slurry waste. Ten ml of the carbonate slurry waste was put into the sealing vial. After standing sample for certain duration, concentration of hydrogen was measured by gas-chromatography. Total amounts of hydrogen gas produced from the slurry was proportionally increased with time. Measured G-value was equivalent to the theoretical G value. This result showed that hydrogen gas generated by water radiolysis in radioactive carbonate slurry waste.
Sato, Junya; Suzuki, Shinji*; Kato, Jun; Sakakibara, Tetsuro; Meguro, Yoshihiro; Nakazawa, Osamu
no journal, ,
no abstracts in English
 -ray irradiation
-ray irradiationNagaishi, Ryuji; Motooka, Takafumi; Yamagishi, Isao; Inoue, Masao*; Matsumura, Taichi
no journal, ,
no abstracts in English